New FDA Record Keeping Rules
As part of the new rules for nutrition labels, the FDA has added requirements for keeping records. Food manufacturers take note: you now have to take notes. You have to make and keep written records to support certain information that is included in your nutrition panels.
Who's Affected
- Manufacturers who voluntarily declare Vitamin E or Folate on their labels.
- People who add synthetic fiber that's not on the FDA's "beneficial" list (like inulin, for example) or separated fiber (like wheat fiber rather than the whole wheat kernel).
- Anyone who adds some kind of sweetener to a product should check to see if they have to keep new records to verify the added sugar content.
Specifics—When and What to Track
Five situations are covered by the requirement. You might have to keep records if:
1. You add non-digestible carbohydrates to your product. A "non-digestible carbohydrate" is one that doesn't meet the FDA's definition of dietary fiber. (They define dietary fiber as “non-digestible soluble and insoluble carbohydrates (with 3 or more monomeric units) and lignin that are intrinsic and intact in plants; isolated or synthetic non-digestible carbohydrates (with 3 or more monomeric units) determined by the FDA to have physiological effects that are beneficial to human health.")
The FDA has a current list of 7 isolated or synthetic compounds that it considers to "have physiological effects beneficial to human health." If your product has any non-digestible carbs that are isolated or synthetic and are not on the list, you'll have to keep records showing the source and amount of "intrinsic and intact" fiber as well as the source and amount of "isolated or synthetic" compounds, which will now be part of total carbs only.
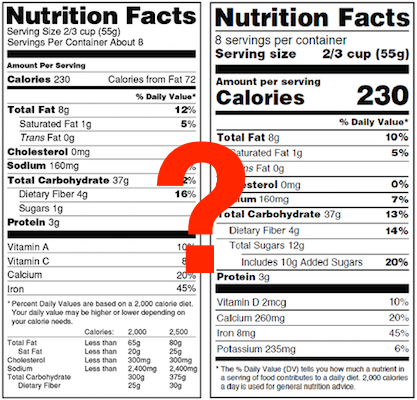
2. Vitamin E is in your product as a mixture of all rac-α-tocopherol acetate and RRR-α-tocopherol. If you choose to declare the Vitamin E, you will have to verify how much of each type is in your finished food.
3. A mixture of folate and folic acid is in your product. Again, if you declare it, you have to make and keep records verifying sources and amounts of each type that are in the finished food.
4. Added sugars and naturally occurring sugars are both in your product. You will need records for the declared amount of added sugars in the food. This requirement covers things like sweetened yogurt or chocolate milk where you have the sugar content of the milk itself plus whatever sweetener you add.
And, finally,
5. Added sugars are declared and those sugars are subject to non-enzymatic browning or fermentation. ("Non-enzymatic browning" refers to the results of cooking methods like caramelization or baking.) This provision applies when the amount of added sugars is reduced through non-enzymatic browning or fermentation. So if your nutrition facts claim that there's less sugar in your finished product than there is in your unfinished (e.g. unbaked) one, you will need records to document the difference and verify the declared amount of added sugar.
Types of Records
The FDA is trying to be flexible and is not requiring specific forms or particular types of documents. Records could be things like the weight of a certain nutrient relative to the weight of a total batch, the results of tests the manufacturer has conducted, the certificate of analysis from a supplier, etc.
No Proprietary Information Wanted
Records submitted to the FDA for inspection only need to show information on the nutrient in question. Information on other nutrients doesn't have to be included. And the FDA wants any proprietary information in records either redacted or marked as proprietary before submitting anything to them as part of a request.
How Long Do You Have to Keep the Records?
You have to keep records for "2 years after introduction or delivery for introduction of the food into interstate commerce." The records also need to be reasonably accessible at each manufacturing facility. The FDA considers electronic records reasonably accessible, and they also consider access within 24 hours reasonable.
Why the Requirement?
Basically, given all their other rules and constraints, the FDA needs a way to determine compliance for a bunch of new labeling requirements that are really hard to measure. So they want manufacturers to help them because, under Federal law, they have no accepted analytical way to do it themselves (there's no way to test for Added Sugars, for example, since they're the same as other sugars).
Bottom Line—Just Do It!
Whether or not that makes any sense, here's something that's clear: "Failure to make and keep records. . .would result in the food being misbranded . . . ." And that triggers FDA warnings and action. So keep those records to the best of your ability (how you do it is pretty flexible), and keep them handy for 2 years.
Let us know if you have any questions in the comments below or email/chat us.