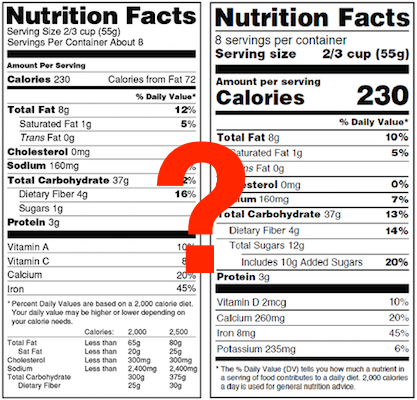
Compliance Dates for the New FDA Nutrition Label Format
The FDA's new rule for nutrition facts panels goes into effect July 26, 2016. The compliance date is July 26, 2018, but smaller businesses with annual food sales that are less than $10 million get an extra year to comply--until July 26, 2019.
EDIT (May 4, 2018): The FDA has finalized and published an extension to the compliance date for the new nutrition labeling rules. The new compliance date is January 1, 2020 for manufacturers with $10 million or more in annual food sales, and an extra year, until January 1, 2021, is provided for manufacturers with food sales of less than $10 million.
EDIT (June 15, 2017): The FDA has announced plans to extend the compliance date for the new nutrition labeling rules. They have yet to decide on the new compliance dates, so for now this is an indefinite extension.
Questions, Questions
We've gone over the changes and dates already, but like a lot of rules, the more you look at them the more questions you have. One of the big ones percolating through food industry circles is "what exactly do the dates mean?" Does the grace period only apply to businesses and products that are already in the market? Do new products and businesses that are created or introduced on or after July 26 of 2016 have to use the new format right away since the new rule will be in effect then? How do you actually, practically, comply?
Our Answer
Here's the best information we have, whether you're already in business or just getting started:
- As of July 26, 2016: You can use the new label format.
- As of July 26, 2018 (or July 26, 2019 if annual food sales are less than $10 million): You must use the new format.
- Between July 26, 2016 and July 25, 2018 or 2019: You can use either the old or the new format. (We'll have both available in ReciPal.)
EDIT (March 23, 2017): The FDA originally said that the new labels had to be on foods that "are initially introduced into interstate commerce on or after the compliance date." That was apparently confusing, so they've changed their thinking and are now pegging compliance to the date the food is labeled.
EDIT (March 23, 2017): So anything that's labeled on or after July 26, 2018 (and July 26, 2019 for manufacturers with less than $10 million in annual food sales) must use the new label. But anything that's labeled before those dates "does not need to be in compliance with the new labeling requirements, and therefore, does not need to bear the new nutrition label." They don't care about where the food is in the distribution chain, only about when you put on the label.
Sooner is Better
It's always a good idea to make the transition sooner rather than later. According to the FDA: "We generally encourage industry to comply with new labeling regulations as quickly as feasible, however. Thus, when industry members voluntarily change their labels, it is appropriate that they incorporate any new requirements that have been published as final regulations up to that time."
So that's our best understanding of the general requirements right now. We'll keep you posted if any other information comes along or anything changes. If you have specific questions or concerns about your situation, you can always contact the FDA. For more general questions, send us an email or leave a comment below.